What is battery? Types of battery, Primary and Secondary cells
Last Updated on August 23, 2024 by Engr. Shahzada Fahad
Table of Contents
Battery Introduction:
Battery– We have been using electric batteries for years, even right now the device you are using has a battery. It really doesn’t matter whether you are using a computer system, Laptop, IPAD, or a Cell Phone, etc. All these electronic devices have batteries. A battery can be as small as the size of a button which you will normally find in the wristwatches and other wearable electronic gadgets.

Or
A battery can be as big as the YUASA CARGO Super Heavy Duty Battery and even bigger e.g. a battery bank which can be as big as the size of a room.

Such type of Heavy Duty Batteries, when used with the UPS, can power up the entire house for hours. You know very well, batteries are available everywhere on this planet ;), now batteries do exist in space as well. Now let’s start the real work.
Without any further delay let’s get started!!!
What is Battery?
The type of devices in which the Chemical Energy is converted into Electrical Energy is called electric cells. When a specific number of these electric cells are electrically connected in Series and Parallel combinations it forms a Battery. Basically each electric cell is made of two different or dissimilar conductors that are immersed in the conducting liquid. So, an electric Battery is such a type of electrochemical device that converts the chemical reactions happening among the electric cells into electrical energy. A battery can have one or more electrochemical cells with external connections called electrodes for powering electrical devices such as UPS, Mobile Phones, Flashlights, and Electric Cars, etc. The electrodes are normally marked with + and – signs.
Types of Battery:
Normally, there are two types of batteries
- Primary Battery
- Secondary Battery
Confused? Let me make it simple. Do you know about the non-chargeable and rechargeable batteries? Non-chargeable battery = Primary Battery and rechargeable battery = secondary battery. Let’s discuss each one in detail.
Primary Battery:
A Primary Battery is the type of battery in which the chemical reaction once happened cannot be reversed i.e the chemical reaction is irreversible. A Primary battery is designed for a single-use and that’s why it is also known as the disposable battery. The Primary Batteries are used once and when discharged these batteries are discarded, as the chemical reaction between the electrodes and electrolyte during the discharging process is irreversible. In more simple words, a primary battery once discharged cannot be charged again. An Alkaline Battery is a type of primary battery that derives its energy from the reaction between zinc metal and manganese dioxide.

These types of batteries you will normally find in portable electronic devices, Tv remote controllers, toys, etc.
Primary batteries, or primary cells, can produce current immediately on assembly. As I said earlier primary batteries are most commonly used in portable devices that have a low current drain.
since the chemical reactions are not easily reversible and active materials may not return to their original forms. Battery manufacturers recommend against attempting to recharge primary cells. Moreover, the primary batteries are cheap, easily available, and easy to use. Mainly carbon-zinc batteries and alkaline batteries predominate the market of primary batteries throughout the world. The Mercury based and Lithium-based batteries are used in situations where very small current, long service life, and very small sizes are required.
Secondary Battery:
The chemical changes inside the Secondary battery are changeable or reversible. A secondary battery is also known as the storage battery. A storage battery or Secondary Battery or a rechargeable battery can be discharged and recharged multiple times using an applied current; the original composition of the electrodes can be restored by reverse current. When an external load is connected with the rechargeable or secondary batteries, the forward reactions take place in the chemical of the battery for delivering electrical power to the load. This process is known as the discharging of the battery. Again when the rechargeable or storage battery or secondary battery is connected with the external power source; the original composition of the electrodes can be restored by the reverse current, which makes the secondary battery ready for the next discharge operation. This process is known as charging of the battery, which you are quite familiar with. As most of the time, we keep charging our laptops, cell phones, etc. Examples include

The lead-acid batteries used in vehicles and

The lithium-ion batteries are used for portable electronics such as laptops and mobile phones. Secondary batteries are expensive when compared with primary batteries. The secondary batteries are preferred to be used where the users are ready to pay the higher initial costs to ensure high current delivery in a consistent and reliable manner. Nickel-cadmium and Lead-acid batteries are mainly used for commercial applications. Nickel-hydrogen battery and Silver zinc battery are the secondary batteries, these types of secondary batteries are used where the performance requirements are crucial than the installation and maintenance cost.
Primary Cells:
A type of cell, that creates an electric current through an irreversible electrochemical reaction occurring in the cell. A primary cell is a battery that is designed to be used only one time. When a primary cell is discharged, it is discarded, and not recharged again like the secondary cell “rechargeable battery”.

A primary cell is basically a galvanic cell, a galvanic cell or voltaic cell, is an electrochemical cell that derives electrical energy from spontaneous redox reactions taking place within the cell.
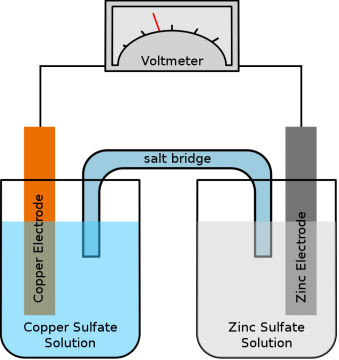
It generally consists of two electrodes made up of different types of metals immersed in an electrolyte. As you can see in the picture above, the two electrolyte solutions Copper Sulfate Solution and Zinc Sulfate Solution are connected through a salt bridge. A Copper Electrode is immersed in Copper Sulfate solution while the Zinc Electrode is immersed in the Zinc Sulfate Solution. Now if you connect the two terminals of a voltmeter with the Electrodes it will show voltage. The voltage produced entirely depends on the type of electrodes and the electrolyte.
Let me give you some examples if the Electrodes are made up of Zinc and Lead when these electrodes are immersed in the Sulfuric Acid solution, it produces voltage around .5 volts.
If the Electrodes are made up of Zinc and Copper, when these electrodes are immersed in the Sulfuric Acid solution, it produces voltage around 1v.
Similarly, if the electrodes are made up of Zinc and Silver, if these electrodes are immersed in the Hydrochloric Acid instead of Sulfuric acid; it will produce around 1.2 volts.
The distance between the Electrodes or changing the size of the Electrodes will have no effect on the produced voltage, but it will affect other things.
A good primary cell consists of the following properties:
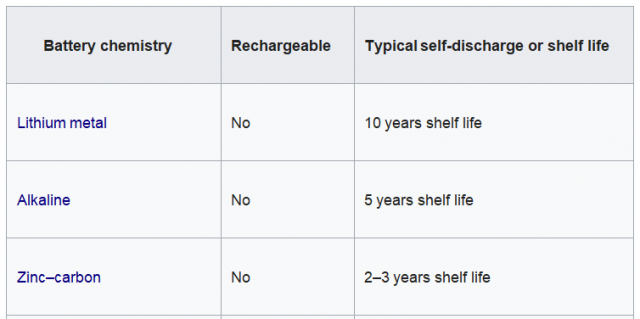
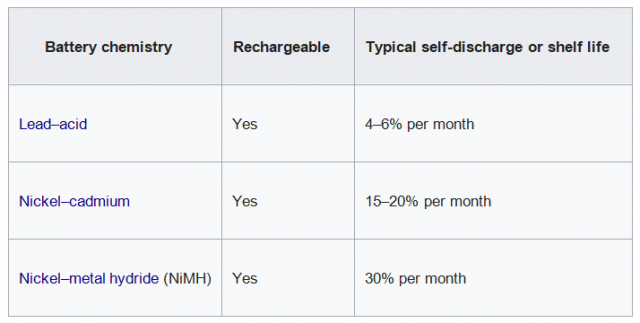
- There should no electrochemical reactions when there is no external load connected, the materials should not be wasted in the absence of electrical load, but this can be an ideal situation because, in reality, nothing is perfect. Batteries Self-discharge, self-discharge is a phenomenon in batteries in which the internal electrochemical reactions reduce the stored charge of the battery without any connection between the electrodes or any external circuit. Different batteries have different self-discharge rates, as you can see in the table given below.
Typical Self-discharge by battery type:
- A good primary cell should produce enough voltage to supply enough current or energy to the load connected.
- It should made up of low-cost materials.
- It’s internal resistance and back EMF should not be greater, otherwise, it won’t be able even to provide low current.
Types of Primary Cells and working:
- Daniel Cell
- Leclanche Cell
- Dry Cell
- Gravity Cell
- Ruben Cell
- Weston Standard Cell
- Zinc-chloride Cell
- Manganese Alkaline Cell
- Mercury Silver Oxide Cell
- Lithium Cell
- Clark Cell
Above are the most famous types of Primary cells amongst which the first 3 types “Daniel Cell, Leclanche Cell, and Dry Cell” are most commonly used.
Daniel Cell:
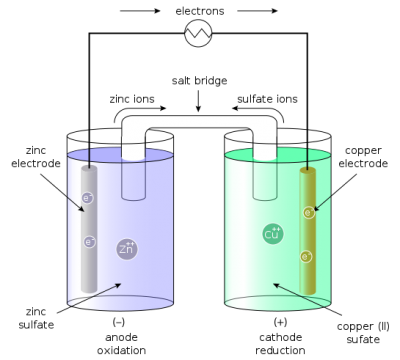
This is a two-fluid cell. The Daniel Cell is a type of electrochemical cell which was invented in 1836 by John Frederic Daniell, consists of a copper pot filled with a copper sulfate solution, in which is immersed an unglazed earthenware container filled with sulfuric acid and a zinc electrode. In Daniel Cell, two types of electrolyte solutions are used as you can see in the diagram above which is a more simplified representation of the Daniel Cell, Copper Sulfate and Zinc Sulfate are used as an electrolyte. Daniel Cell has two electrodes which are made up of Copper and Zinc. These electrodes are immersed in electrolyte solutions.
The cathode or the positive electrode is made of copper plate, as the copper electrode is immersed in the copper sulfate solution due to which there is no polarization. The Daniel Cell is designed for those circuits which remain close, when the circuit is opened or disconnected, the electrodes are wasted. The Daniel Cell can produce around 1.1volt, practically this cell has very limited use. The Daniell cell was a great improvement over the existing technology used in the early days of battery development. A later variant of the Daniell cell called the gravity cell or crowfoot cell was invented in the 1860s by a Frenchman named Callaud and became a popular choice for electrical telegraphy.
Working of Daniel Cell:
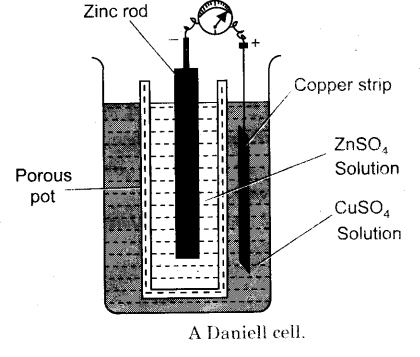
Now we will discuss how a Daniel Cell works. As you can see in the picture above in a Daniell cell the Zinc electrode is slightly bigger than the Copper electrode. When the Daniel Cell is connected in a circuit, let me remind you this is a closed-circuit cell, the Zinc electrode with its electrolyte solution starts converting into Zinc Sulfate. The metallic copper from the Copper sulfate solution starts accumulating at the Copper electrode which is the Cathode, due to this the weight of the copper electrode keeps increasing while on the other hand, the weight of the Zinc electrode keeps reducing.
This is why while making the Daniel Cell the Zinc electrode is made bigger and the Copper electrode is made thinner. The slow reduction of the Zinc electrode or plate is called the local action. The Daniell Cell internal resistance is from 3 to 5 ohms, and for one cell, 9 Ounce Copper sulfate and 2 Ounce Zinc sulfate is needed. For the best results, the circuit is kept closed if the circuit is kept open the Copper sulfate will slowly start mixing with the Zinc sulfate and the cell will become a waste. The Daniel Cell is used in relay signals, Fire Alarm systems, Telephone exchanges and all types of other close circuits. Presently, the storage batteries or Secondary batteries are more popular.
Leclanche Cell:
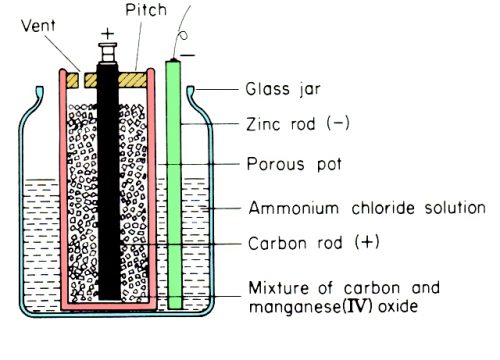
Leclanche cell is the most popular type of the Primary battery, because the Leclanche cell is the basic shape of the most commonly used Dry Cell. This cell was constructed by Georges Leclanche in 1865. In this cell, Anode is a carbon rod placed in a porous pot containing a mixture o powdered carbon and manganese dioxide. Manganese dioxide is an oxidizing agent and acts as a depolarizer. The carbon powder is used to make the manganese dioxide conducting or to increase the surface area of the carbon electrode. An amalgamated zinc rod is used as the negative electrode “Cathode”. The porous pot and the zinc rod are placed in a glass jar containing the solution of ammonium chloride (10 parts water and 1 part of ammonium chloride) which acts as an electrolyte. The mouth of the jar is painted black so that the ammonium chloride may not creep the walls of the glass jar. When zinc is mixed in the electrolyte it is converted into the zinc ions, as a result of this chemical reaction the electric potential is generated.
Dry Cell:

Dry cells are the types of chemical cells or batteries which are used to store electricity. The dry cell is the developed form of the Leclanche cell. A Dry cell is a compact form of Leclanche cell. A Dry cell as the name implies, in reality, is not a Dry cell. The electrolyte of the Dry cell is mixed with the absorbent inactive material which makes a paste that is packed inside the containers made of Zinc. The entire cell is sealed in a watertight container to protect it from evaporation. Due to this it can be easily transported and can be used in any position. These cells are easy to carry due to lightweight and small size. Practically the Dry cell has replaced the other types of the primary cells.
The container of the Dry cell acts like the –Ve electrode or Anode which is made of Zinc. Normally these are of the cylindrical shape. Inside the Zinc container, there is a layer of non-conducting material, for example, Blotting paper or Plaster of Paris which is saturated with electrolyte, a moist paste of ammonium chloride. The Carbon rod which is the Positive electrode is surrounded by the Manganese (IV) oxide. At the top and bottom of the cell, there are metal contacts for easy connections.
A common dry cell is a zinc-carbon cell, sometimes called the dry Leclanché cell, with a nominal voltage of 1.5 volts, the same as the alkaline cell (since both use the same zinc–manganese dioxide combination).
Dry cells are used as batteries in toys, calculators, clocks, flashlights, and many household appliances.
Dry cells should not be used as the main source of power in devices that operates most of the time, because these cells will discharge soon. Dry cells are ideal for intermittent services. For examples when a dry cell is used in a TV remote it lasts for more than a year due to the less usage.
Secondary Cell:
A type of Cell in which the physical and chemical state of the electrodes and electrolyte can be restored through charging is called a Secondary Cell. A secondary cell can be charged and discharged so many times, while the primary batteries are supplied fully charged and discarded after use, after a long series of experiments on the primary cells, some type of the cells are invented in which the chemical reactions can be reversed with the help of electric current through the charging process, such cells are called Secondary cells.
In case if High voltage and High current are needed, the secondary cells can be connected in series and parallel combinations to achieve the desired voltage and current, such type of the battery is called a storage battery or Accumulator. A secondary cell is also known as the storage cell.
The voltage of a secondary cell depends on the type of electrolyte and the material of electrodes. The capacity of a secondary cell depends on the area of the plates used, larger the plates area, greater will be the cell capacity. These cells are connected in different styles to make the storage batteries. When these batteries are discharged, then specially designed DC power supplies are used to charge these batteries again, this process is called the battery charging process. Charging and discharging is the main feature of the Secondary cells. These batteries are used in Grid Stations, telephone exchanges, Radios, Transmitters, telegraphs, military equipment, Cars, Buses, Air Planes, with UPS inverters, DC room coolers, DC ceiling fans, etc. These batteries can deliver hundreds of Amps for a short period of time and can deliver low current for long periods of time.
Self discharging of the Secondary Battery:
Types of Secondary Cell:
- Lead Acid Cell
- Nickel-cadmium alkaline cell
- Nickle Iron Alkaline or Edison cell
- Silver Zinc Cell
Discover more from Electronic Clinic
Subscribe to get the latest posts sent to your email.