Semi Conductors Theory, Valence Electrons and Covalent Bonds
Last Updated on March 21, 2022 by Engr. Shahzada Fahad
Table of Contents
Semi-Conductors Introduction
About 20 years ago, vacuum tubes of different sizes and shapes influenced the entire world electronics industry due to its myriad benefits. In the present era, the semi-conductor technology has created a revolution in electronics industry. As compared to vacuum tubes, semi-conductor materials are extremely small in sizes, least heavy, simple from manufacturing viewpoint, stronger and more effective. Further, there is neither heater loss nor they require time or heater for warming up. Due to these advantages of semi-conductors, the existence of vacuum tubes came to an end in electronics market (with the exception of few places e.g. picture tubes) and semi-conductors have successfully taken their place. The study of semi-conductors is very important for knowing about semi-conductor diodes, transistors and more advanced ante grand circuits. The application of semi-conductors in computer technology, industrial controls, medical technology, space technology, life sciences, chemistry, physics, politics and above all war technology has achieved tremendous popularity. Semi-conductors are now successfully been used in different appliances ranging from few small diodes to more power controlling silicon control rectifiers. Semi-conductor chip or anti grand circuit (I.C) chip has gained astonishingly huge popularity in small gadgets e.g. calculators, watches, radios, hearing devices, video cassette recorders, tv, receivers, microcomputers, several equipment and control systems.
Semi-conductor Theory
Materials can be divided into following 3 categories on the basis of flow of electrons or resistance in the way of current.
- Material which offers severe resistance in the flow of current (106 ohmsxcm) are called insulators.
- Material which offers little resistance (10-6 ohmxcm) in the flow of current, are called conductors.
- Materials which offer medium resistance in the way of flow of current or flow of electrons (102 ohm x cm) are called semi-conductors.
The meaning of semi is half, partial, between or intermediate. Semi-conductors are solid materials (elements) electrical properties of which are located between conductors and insulators i.e. these materials are neither good conductors nor good insulators, rather its electrical properties lie between these two. In other words, materials the conductivity of which is greater than a good insulator and lesser than a good conductor, or such solid materials, the resistivity of which is greater compared to conductors and lesser to insulators, are called semi-conductors e.g. germanium and silicon.
A semi-conductor material is one whose electrical properties lie in between those of insulator and good conductor e.g. germanium, silicon etc.
Remember that the dependence of entire electronics industry is on this class of material these days as all electronics equipment and anti-grand circuits are manufactured from semi-conductor materials. The material which is most widely used as a semi-conductor is silicon. However, carbon, germanium, gallium arsenide (Ga A5) are also used in several important appliances.
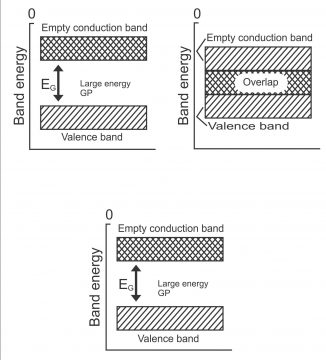
From viewpoint of energy band, semi-conductor is defined as materials, which on room temperatures consist of nearly empty conduction band and partially filled valence band. Between these two bands, a small energy gap (approx. equal to (IeV) exists. The resistance of such semi-conductors reduces with an increase in temperatures. Small energy gap means that very little energy is required for valence electrons of this material going to conduction band. Some valence electrons go to conduction band at room temperature through absorbing suitable energy, thus they become free electrons.
At 0⁰K, no electrons exist in the conduction band of a semi-conductor, while its valence band is completely filled. This means that a very small of germanium or silicon acts as a fine insulator at absolute zero temperature. However, the width of forbidden energy band gets less due to an increase in temperature. In this way, some of the electrons enter conduction band. In other words, conductivity of semi-conductor band increases due to an increase in temperature, which means that these are capable of negative temperature coefficient. In figure1, the energy band of the insulator, conductor and semi-conductor has been reflected.
Valence Electrons
We know that the number of electrons present in the outer shell or orbit of an atom is called valence electrons. The most stable shell of an atom is one, the last shell of which is complete, i.e. in the last shell of which there are maximum 8 valence electrons. Due to the completion of shell of last atom, neither any electrons enter from outside nor its electrons can transfer to some other atom. Thus, this atom becomes strong. The electric properties of any material also depend on the number of valence electrons present in the last shell i.e. the electric properties of materials can also be judged by the number of valence electrons present in the last shell. The definition of semi-conductor from the viewpoint number of electrons in the last shell (ring) of an atom can be done as under. Semi-conductor is such an element or material, which has 4 electrons in its last shell i.e. carbon, silicon, germanium etc. (Silicon was discovered in 1823 whereas germanium in 1883). (Semi-conductor materials typically have four electrons in the outermost valence ring).
Energy Shells
Paths surrounding an atom on which electrons move, are called energy shells. The outermost orbit of an atom is called valence band. Atomic number of silicon is 14, i.e. a silicon atom consists of 14 protons. It means that the number of electrons moving around different shells (rings) of a silicon atom is 14. (As the number of electrons and protons in an atom always remain equal, therefore, every atom is electrically neutral. The division of electrons in different shells or orbits is reflected by 2n2formula, the division of electrons in a silicon shell is done in such a manner, that the first orbit has 2 electrons, 2nd orbit has 8 and 3rd or last shell has 4 electrons in it. This division has been shown in figure2(a). Similarly, a germanium atom has 32 protons and 32 electrons. The rotational division of these electrons in various orbits has been shown in figure2 (b)
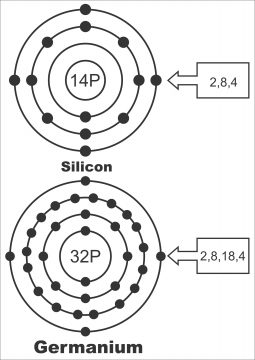
It becomes obvious from above detail that electric properties of some materials can be identified through number of electrons found in an atom’s last shell valence orbit. For instance, if a material has 8 electrons in the last shell, it functions as an insulator. Conductor consists of just a valence electron. While 4 electrons exist in the last shell orbit of a semi-conductor. Semi-conductors have the following properties:
- Its electric properties are found between conductor and insulator
- It has greater resistance compared to conductor, while less resistance as compared to insulator
- It consists a nearly empty conduction band and a nearly filled valence band. And a small energy gap exists between these two bands.
- Its resistance diminishes with an increase in temperature, i.e. its temperature is coefficient negative.
- Its conductivity is better than a good insulator, whereas less than an good conductor
- The number of valence electrons in its last shell remains 4
Energy Levels
If we start lifting a stone from the ground, the higher its height from the ground, the more will be the amount of energy going to be consumed to lift it. The stone absorbs this energy as potential energy from the earth. If we let the stone-free, it will fall to earth. And the energy, which was used at the time of picking up the stone, will emit upon its getting to the ground.
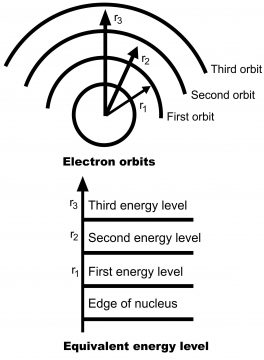
The energy levels of electrons present in an atom, can be explained with the help of an example. Suppose, a silicone atom has been shown so magnified in size, that we can easily see each of its orbit, as has been shown in the diagram 1.23. An electron requires energy for moving from one orbit to the other, so that attraction of nucleus could be controlled. When an electron moves towards a higher orbit, it gains potential energy in order to overcome nucleus energy(through which it has been firmly bonded with a nucleus). If electrons are allowed to return to its original orbit, it emits such absorbed energy.
Curved orbits have been revealed in diagram (a), while in diagram (b) equivalent energy levels have been reflected. The first is called the first energy level, 2nd the orbit is called 2nd energy level. The bigger the size of orbit, the greater will be the energy level. If atom is provided energy through some external source, its electrons move to higher energy levels through absorbing energy. The electrons located near the nucleus, have small energy, while the electrons away from the nucleus, have greater energy. Thus, as the difference of an atom from the nucleus increases, its energy level also increases.
Covalent Bonds or Atomic Bonding
We know that there are 2 electrons in the 1st orbit of a silicon atom, 8 in the 2nd while 4 in the 3rd orbit. This phenomenon is correct for a single isolated silicon item. However, when several silicon items combine together, they assume a solid form. These atoms automatically arrange itself in such a manner, that they turn into a crystal. The solid structure which comes into existence through covalent bonding as a result of meeting of atoms, is called crystal. If every silicon atom connects with other silicon atom in such a way, that the number of electrons in its valence orbit become 8, this atom becomes chemically stable. That’s when 4 electrons present in the last orbit of semi-conductor atom, connect with 4 electrons of another semi-conductor atom, the final shell gets completed through their mutual combination. This operation is called covalent bonding. When valence electrons complete a covalent bond through combining with other electrons, further electrons can neither be included nor emitted from the last shell of semi-conductor atom, thus, these materials assume a crystalline shape and thus they become insulators from conductors. One or more than one valence electrons between two atoms participate in this operation, in which each atom tries to complete its outermost shell or final orbit. The process of covalent bonding in semi-conductor materials (e.g. silicon or germanium) has been depicted through 2 processes in the c. Only valence electrons have been shown in the diagram.
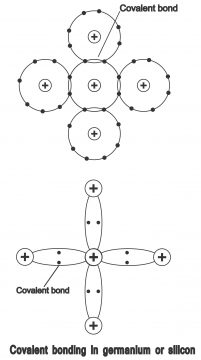
It has been shown in the figure that each of the 4 electrons present in the last shell of the silicon atom, develops a covalent bond through trying and combining with electrons of other surrounding atoms, thereby, increasing electron numbers to 8. Such bonds can be broken through providing suitable energy on solid. Electrons, which get free from breaking of bond i.e. emits from the last shell of atom, creates an empty space or vacancy, which is known as positive hole. Thus, breaking of every bond result in the creation of 2 charge carriers, one of which is electron and the other one being, hole. A silicon atom manages to have 8 in its last shell, which also can be explained that as per figure 3, when 4 extra electrons are present around a silicon item, every closer atom, provides an electron to the central atom i.e. other 4 atoms present around the central atom, provide one electron each to the central atom. Thus, through acquisition of four additional electrons by the central atom, the number of electrons in the valence orbit becomes 8.Actually, electrons cannot stay in an atom for too long and they keep on a mutual operation with adjacent atoms. As the number of electrons in a valence orbit, cannot exceed 8, therefore it is known as filled or saturated when it has 8 electrons in it. Just like silicon, an isolated germanium atom also contains 4 electrons, when germanium atoms combine mutually, they get a solid shape. Thus, they also assume the form of a crystalline structure, as shown in the figure4
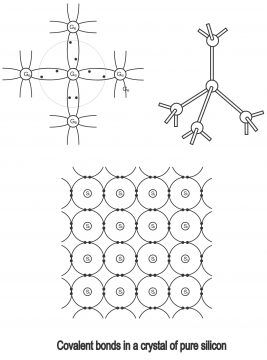
In figure 5, covalent bond produced in a pure silicon crystal has been depicted
Free Electrons and Holes
When ambient temperature (temperature found in air and surrounding atmosphere) increases from absolute zero, i.e. when temperature is greater than 460⁰F, silicon or germanium crystal atoms vibrate through produced heat. The mechanical vibration of these atoms increases with an increase in ambient temperature. If a hot crystal is held in hand, heat is felt in hand due to atoms’ vibration. Due to this thermal energy or too much vibration of silicon atom, one electron separates from valence orbit (fig5). This separated electron moves to a higher orbit through getting more energy. These electrons become free after getting to the bigger orbit as the attraction of nucleus on it becomes virtually negligible (electron, on which nucleus attraction is so minimal, that it can easily move from one atom to the other, is called free electron), due to which free electron moves freely within the entire crystal. A vacancy is generated in valence orbit, as a result of movement this electron from valence orbit to a higher orbit, which is called hole. This hole acts as a positive charge, because it can attract any nearby electrons towards it for entering the hole. The number holes produced and quantity of free electrons is equal in a pure silicon crystal due to the effect of thermal energy. These free electrons move freely inside the entire crystal. There is a positive charge in hole, while there is a negative charge on electrons. Hole attracts electrons towards it, due to which these free electrons drop and fill the hole. The operation, through which free electrons and hole get together, is called recombination. Any moment, following operations take place inside a silicon atom.
- Some free electrons and holes are produced through thermal energy
- Some of the produced free electrons and holes remain recombined.
- Some free electrons and holes remain in the middle condition, i.e. they have not yet been produced, and they cannot recombine.
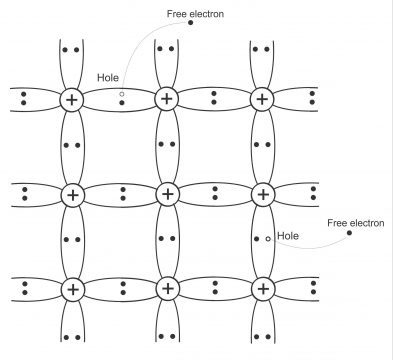
Intrinsic germanium structure after application of heat showing two broken covalent bonds, in two free electrons and holes; “electron-hole pairs”
Energy Bands
We know that electrons originated in the outermost shell of an atom, are called valence electrons. The orbital energy of such electrons is maximum, while their binding energy is minimum. The energy band, wherein valence electrons exist, is called valance band. This band does not vacate, rather remains fully or partially filled with electrons. (as shown in figure 6)
Electrons which tend to emit from the valance band, are called conduction electrons and the band where conduction electrons subsist, is called band. Conduction band take place after the valence band. This band can be emptied completely partially or it may remain full. Electrons moving freely inside the conduction band, pass current through a solid, therefore they are also called conduction electrons.
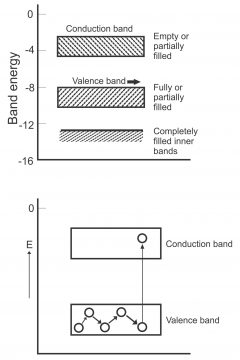
The gap between the valence band and conduction band is called the forbidden energy gap. For moving from valence band to conduction band, an electron has to pass through a forbidden energy gap, for which it requires energy. When a valence electron absorbs some suitable energy, it enters the conduction band. (figure7). Electrons present in the conduction instead of moving back to the valence band may enter other conductance bands easily. A huge amount of energy is required to be released from conduction electrons for moving to valence bands, therefore they drop again in the valance band and return back to the conduction band after the revival of energy.
When an electron emits from the valance band, a covalent bond breaks, therefore, a positive hole develops at the place of emission of electrons. This hole attracts electrons freed from the nearby atom, and electrons getting free, through breaking covalent bond moves towards hole. Thus, hole fills due to the combination of hole and electron. Remember that all holes are filled through these electrons, which move without crossing prohibited energy gap of these nearby atoms.
In other words, when an electron in valence orbit gets suitable thermal energy, it moves or jumps from the energy gap to the conduction band (At absolute zero temperature, there are no free electrons in a silicon crystal. The empty places in the middle portion of an energy band are called forbidden gaps because they show energy levels or unstable orbits) Figure 1.30 Suppose that a covalent bond breaks at “A” due to the emission of electrons. In this way, a vacancy or positive hole is produced in valence band. The electron of a nearby atom moving from B to A jumps into hole A and fills it. In this way, the actual hole is filled, however, a new hole produces on B. Now an electron jumping from C fills a hole produced on B, thus this process moves on.
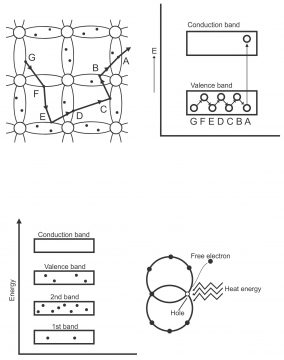
Different energy bands found in a silicon crystal have been shown in figure 7(a) at absolute zero temperature and above absolute zero (when electrons move from valence band to conduction band and holes are produced in valence band) shown via figure(b). When voltages are provided by means of battery parallel to a piece of silicon, free electrons nearby in the conduction band, start moving easily towards positive end of the battery. The movement of free electrons in a semi-conductor material results in the flow of electron current, while the flow of holes is towards the opposite side of the flow of electrons, which is called hole current (details are going to be provided in the coming pages)
For electronics and programming-related projects visit my YouTube channel.
Previous article: Pentode and Next article: Types of Semiconductors
Discover more from Electronic Clinic
Subscribe to get the latest posts sent to your email.



This is something that… every electronics engineering should know…