Liquid Crystal Displays Technology Fully Explained
Last Updated on September 12, 2021 by Engr. Shahzada Fahad
Table of Contents
Liquid Crystal Display, Overview:
Today we’re going to talk about things that are not quite crystalline but definitely not amorphous as well and we’re a start off by talking about liquid crystals. Liquid crystals are particularly important because they interact with light in interesting important ways. Specifically they can change the polarization of light and so this is basically an image of shining polarized light through a film of liquid crystals and you can see it some parts the light is basically rotated and slowed, some part of the light is cancelled out because it’s rotated and it’s rotated an angle that’s cancelled out by the polarizer that is between the film and where we’re looking at it. So that might seem like a little bit of an academic and not an interesting thing at all until you remember that a lot of technology is based on liquid crystals so a liquid crystal display is basically a something like a monitor or TV where we use a thin film of liquid crystal materials to turn on and off each pixel and we do that specifically by changing their internal structure, changing how they’re oriented so that it affects how they interact with polarized light.
So we’re going to start talking about what these materials are. We are gonna basically focus on some definitions about what are liquid crystals were – some of the different kinds of liquid crystals and we’re going to talk about short and long range order in these systems.
So in order to get started, let’s think about how we describe these different systems before An atomic crystalline solids. So see, there’s things like metals or ionic solids the individual components of that crystal structure are atoms so in this case this is an ionic solid so there might be some cations and some anions but the individual components are atoms and the bonding between the principal components is strong primary bonding and so in this case that might be ionic bonding. In other cases we might be talking about covalent bonding or metallic bonding but the point is that this is very strong primary bonding so bond enthalpies can be quite strong between individual atoms.
So let’s move on to think about a molecular crystalline solid and so what do I mean by a molecular crystalline solid, in this case the individual components aren’t atoms but they’re molecules and so this is an example of ice this is one crystal structure that water model molecules can take. Keep in mind there’s not just one kind of ice depending on the temperature and the pressure that you’re at there are dozens of different crystal structures that have been observed or proposed for ice but the important thing here is that the individual components aren’t atoms anymore but rather they’re molecules and so that means that the bonding between these principal components tends to be secondary bonding, so in this case there are relatively strong hydrogen bonds. If the molecules that we’re talking about are or something like methane then it could be a weaker form of secondary bonding, but it in general it’s a lot less strong than the primary bonding in the atomic solids that we were thinking about before.
So the final question I have here is what are the internal degrees of freedom and by that I mean how much freedom or flexibility does each of these molecules have. Can it have translational degrees of freedom so could it be positioned there or maybe a little bit to the left or maybe a little bit to the right or does it have rotational degrees of freedom, so is this orientation exactly defined or could this be free to rotate around some particular axis and so in this molecular crystalline solid we still have zero internal degrees of freedom because there’s no translational freedom. The positions of each of these molecules are relatively are fixed in this unit cell and there’s no rotational degrees of freedom either each of these angles is determine and the water molecule essentially is frozen it’s stuck in that one position so this is a crystalline solid but a crystalline solid that’s made of molecules as opposed to made of atoms. so this is this is still very much what we’ve been talking about previously this is a crystalline material we would use the same terms unit cells lattice translation vectors to describe this solid.
Now, this is different from what we’re gonna start talking about in this class and these are called liquid crystals and I know the phrase might be a little bit confusing because we see that word crystals again but the important thing here is that they’re liquid crystals and so we ought to talk about what does that mean and let’s ask the same question. So what are the individual components in liquid crystals ultimately they’re still molecules, so the individual composer still molecules but we call them something different we call them mesogens so each of these long ellipsoids it represents a molecule but because as part of a liquid crystal we call it a mesogen and that’s the basic building block of a liquid crystal. the bonding between mesogens again is relatively weak secondary bonding so that means that they there are not these stronger more rigid primary bonds but that gives it the property that now we can start to introduce internal degrees of freedom and I’m just going to say greater than zero or yes at this point and we’re going to talk about these different liquid crystal structures, because they have different numbers of internal degrees of freedom but just in general you can see that you know for example, in this pneumatic crystal there is some translational freedom you know if I look at one molecule and I look at the position of another molecule relative to that this one is not fixed in a well-defined rigid point in space there’s some variability in terms of where that molecule could be, so there’s some translational freedom rotational freedom you know maybe these things are free to rotate around their long axis but less free to rotate around axis that’s perpendicular to long the long axis so greater than zero internal degrees of freedom and the answer that question is what we’re gonna focus on for a lot of the rest of the lecture today.
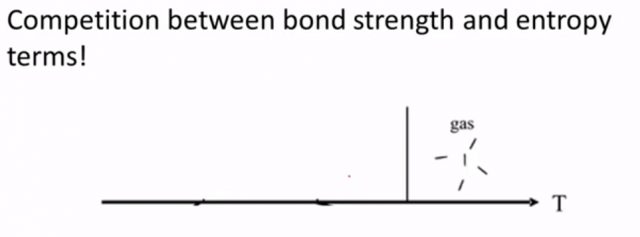
So building up you know another way to think about these things is that liquid crystals they occupy this space somewhere between liquids and crystals and it’s because there’s a competition between bond strength, so how rigidly fixed is this position between one building block and another and an entropy term so that we know it at higher temperature there tends to be more disorder in this system and liquid crystals are kind of somewhere along this spectrum. So if we start off at a high temperature you know things could be a gas a very volatile phase and so there’s a lot of disorder in this system.
As we start to cool down you know gases will condense they can form liquids and so they’re relatively more compact but there’s still a lot of disorder and then traditionally you know we think at some temperature if we get low enough that liquid will crystallize into a crystalline solid and in a crystalline solid the terms related to bond strength tend to win out over these temperature based entropy terms and so that means that there’s not enough energy in the system to overcome the localized potential energy well that is fixing each of these you know units in place liquid crystals are kind of somewhere in between. You know they’re somewhere in between the liquids and the crystals and so we can say that they have a little bit less disorder than liquids so in this particular case things seem to be all relatively lined up in a particular direction but they’re not quite as ordered as a traditional crystal is and so again there are different kinds of liquids and we’re going to talk about the differences between these different types surely.
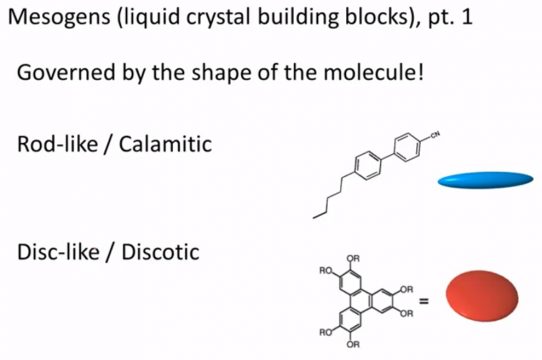
The very first level we can classify liquid crystals by the shape of the molecule and it’s important to note that the shape is what gives it some degree of order so things tend to for example if we have a rod like structure a long axis in a molecule this is something that’s relatively rigid and so it wants to maintain a rod you tend to have these kind of molecules lining up with respect to each other. Similarly, if I have a flat or planar molecule it might make something that looks a lot more like a disc and discs will tend to stack on top of one each of each on top of one another something like this so we can classify these things into rod like or Calamitic types of liquid crystals or discs like or Discotic I tend to just use the simpler terms rod like or disc-like.
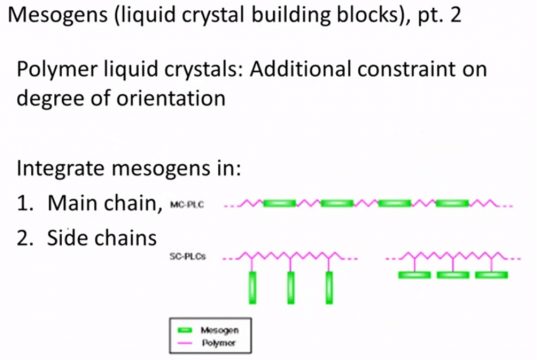
We can also have polymer liquid crystals and so liquid crystal again there’s these individual building blocks the things that we called mesogens in a polymer a liquid crystal mesogens are either in the main chain of the polymer or they’re appearing as side chains along a polymer backbone and these again have unique properties because now the position and how these mesogens can stack is constrained by this backbone polymer itself.
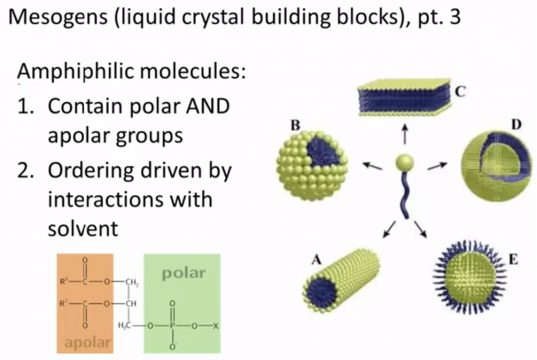
And finally there’s another pretty unique class of mesogens that we’ll deal with later and these are called Amphiphilic and so amph means both and philic means liking so they’re they’re molecules that like two things and the two things that they like are a polar solvents. So a polar solvents are things like hydrocarbons and oils and polar solvents are things like water that have strong dipoles and so because they have this sort of split personality situation when you put a bunch of AMPH you feel like molecules into one kind of a solvent or another they tend to organize themselves so that the tail or the head the part that tends to interact with one kind of solvent or another is exposed to the solvent that it likes and a perfect example of this is a lot of detergents and soap molecules the way these work you know we think about the solvent that we use them in, isn’t water you’re washing your hands and you put some of these and fulfilling molecules well they’ll tend to make you know little spheroid all shells around oil droplets and particles and then they’ll be washed away in the solvent and so the reason detergents and soaps like that work is that they’re picking up oils and residues on your hands or on clothing and they’re allowing it to be washed away in the solvent.
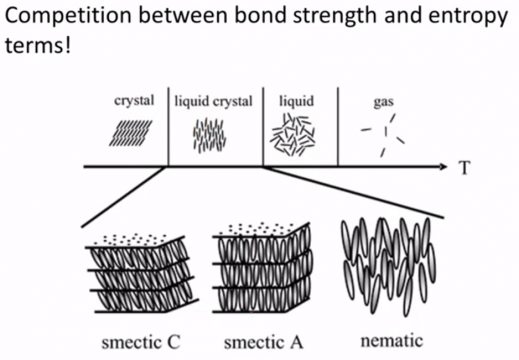
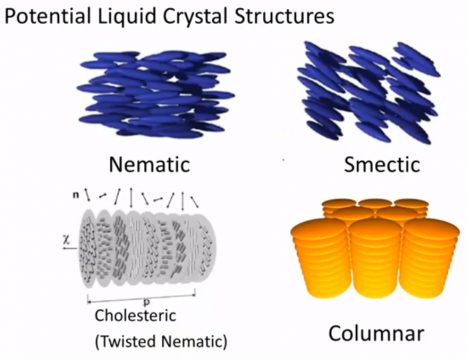
So now we’re gonna talk a little bit more about these different classes of liquid crystals and the two main ones are Nematic and Smectic, there’s also Cholsteric liquid crystals these are these are also referred to as Twisted Nematic and columnar and liquid crystals.
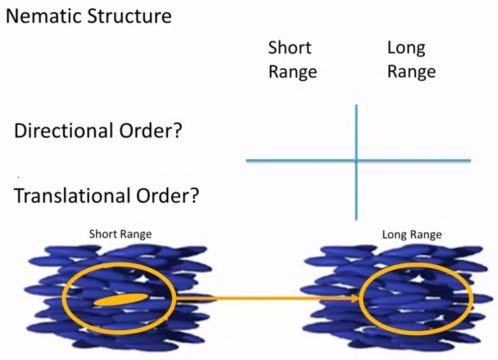
And we’re gonna ask these same basic questions of each of them. Do they have directional order? do they have translational order? and we’re gonna focus on both sort of a short-range degree of order. So the molecules that are immediately surrounding that original molecule and long-range order so think of a molecule that’s about ten to a hundred molecule lengths away from our central point, do we still know something about the or the position or the orientation that molecule in that same place so Nematic structures.
The first question let’s think about short-range order first. Do they have directional order? and do they have translational order? and what do we mean by these terms? so directional order is if I think about the molecules surrounding this initial central molecule are they all pointing and aligned in the same direction and so again we have to be a little careful when we answer this question, because it depends on what rotational axis we’re talking about. So they all tend to be pointing in the same way so they’re kind of lined up and so in that sense if I think about an axis that’s perpendicular to the screen the rotation angle is pretty darn close between all of these nearby molecules, on the other hand if I think about a rotational axis that is along the long axis of the molecule well they’re there they’re relatively free to rotate around that rotational axis and they’re kind of unperturbed by the surrounding molecules and so this is an one where it sort of depends, I’m gonna say yes / no yes because if I’m looking at axes they’re perpendicular plane again all these molecules are sort of pointing left to right but they are free to rotate around that central axis. Do they have translational order? so let’s think about when we were talking about amorphous solids we know that there is always some degree of short-range order if I see you know a molecule here it’s gonna have nearest neighbors that are gonna be you know some pretty well-defined distance away and that’s just because of the shape of the molecule and the surrounding molecules. So there is definitely short-range translational order but by the time I get to a much greater distance away we’ve lost that translational order so I can’t tell you anything about the exact position of this particular mesogen in with respect to that starting point if it’s if it’s some number of molecule lengths away directional order again they kind of have this overall direction that they’re pointing in and I think really we should. We should focus on this one particular aligned direction usually we don’t care too much about rotation around that elongated axis, so you know there’s an argument that there under some circumstances you could say they’re free to spin along their long axis but really the thing that we’re focusing on overall is the orientation of this particular rod.
So we’re gonna go out and talk about the others, but I want to pause and I want to bring back up something that we had learned when we were dealing with amorphous materials and that’s this idea of a pair distribution function. So the pair distribution function – grabs the probability of finding a mesogen in a particular position and one thing that’s useful is to schematically draw the pair distribution function in two directions either perpendicular to the principal director so perpendicular to the direction that these are pointing or along the right should parallel to, it so we’re gonna do perpendicular. First let’s think about what does that pair distribution function look like if I were going to draw it perpendicular to this orientation that the mesogens are lined up I’m gonna give you a second to think about it I’m gonna go ahead and might draw my axes while we’re doing this so you know usually we say this is G of our there’s a probability distribution and so one is sort of the average probability of being aligned in particular direction and this is a radial distance and so we know just like other molecules there’s some minimum position that these mesogens can’t be on top of all and each other and then there’s gonna be some average nearest neighbor distance and that’s gonna be the the maximum of this first peak and then there’s a little bit of an exclusion zone after that and then I have my second nearest neighbor maybe my third nearest neighbor but after about that we’re gonna hit our correlation length. This is Chi the Greek letter guy correlation like that remember that’s the distance after which we don’t have any additional information about where the position of the next molecule would be and so if I’m looking at an in a perpendicular direction my plot might look something like this now I’m gonna change colors here and draw the pair distribution function along the principal director and what’s it gonna look like in that direction. One way to think about this is how is it going to vary from what I’ve just drawn. do we have any additional translational order along this particular direction the answer to that does no oops where we also are you know relatively disordered along the principal director distance direction but there is one thing different and that’s just this nearest neighbor distance is going to tend to be a lot longer, along that distance than it is perpendicular to that direction, so if I was going to draw the pair distribution function and maybe I’ll do it on the same axis you just it would just be stretched out a lot more I would still have a first nearest neighbor a second nearest neighbor or maybe a third nearest neighbor and then we would sort of flatten out. So in general these share a lot of characteristics but because the molecules are elongated it’s stretched out what I’m looking along that one that second direction so this is my overlap so this is the hard sphere radius essentially and nearest neighbor direction along the what we call the principal director that’s basically the vector that is lying along the direction that all these mesogens are pointing.
Twisted Nematic Structure:
next we’re going to think about something called a twisted nematic structure and in many cases this behaves relatively similarly translational order again we’ll have some short-range translational order but we have no particular long-range order directional or the only thing that’s different now is that instead of all of these rod like liquid crystals pointing in one particular direction and that direction staying the same. They tend to twist so the principle director is changing an orientation and the way it’s pointing is a function of some distance soon so this shown as P here and you can in fact you can describe the periodicity in terms of some sort of a sinusoidal function and so we could say that the you know the spacing between or the distance that takes to do a full rotation is given by this two pi Z over lambda term. So in terms of directional order coming back here, they still have short-range order and I’m not going to talk about the fact that they could be rotated around that axis. let’s think about the way that the director is pointing they’re definitely a short-range order there definitely is some long-range order but now that long-range order is not static the direction rather than being fixed over space, the direction and the molecules are pointing tends to twist over space and that’s why these are called twisted nematic structures.
Smectic Structures:
The next class is called smectic structures and these are a little bit different and the reason that they’re different is because they have some additional translational order. So again directional order they tend to be pointing in the same direction as their surrounding molecules, if I go 10200 molecules away they’re still kind of pointing in the same direction, they could be pointing perpendicular to these layers or they could be tilted, so that’s the difference between smectic AR smectic si but the biggest difference between smectic structures and if we go back to Nematic structures is that smectic structures have these layers that we don’t see in the Nematic structures so I could call this layer 1 2 3 and the molecules tend to fall in one of those layers and so what that means is that you know while yes they have some trend sort range translational order they also have some longer range translational order because they tend to fall in a layer although that is only in one direction so if I think about a direction perpendicular to the layers let’s call this X then I could tell you with some certainty positionally where these molecules are but if I think about another direction let’s say a direction within a layer call that y-you don’t have any additional information in terms of the long-range packing of these molecules so they’re restricted to layers there’s some degree of translational order but they are disordered within a particular layer and that’s what this no refers to.
Liquid crystal display,liquid crystal displays,smectic structure,nematic structure,liquid crystal displays,
Discover more from Electronic Clinic
Subscribe to get the latest posts sent to your email.








explore sora solutions services, a Swiss-based Information Technology company. Learn more about our complete hardware and software solutions.
Ecommerce software will encourage customers to use your simple interface to place orders for your products and service. The best ecommerce software will also allow you to simplify your tracking and purchase management through features implemented on the administration side of the site. competitors pricing